CURING LIVER DISEASES USING HUMAN CHEMICALLY INDUCED PLURIPOTENT STEM CELL-DERIVED FUNCTIONAL HEPATOCYTES
2026-04-29
The liver, the largest internal organ in the human body, plays a central role in regulating virtually all metabolic processes. Liver dysfunction leads to a spectrum of diseases that severely disrupt human metabolism, which account for approximately 2 million deaths per year worldwide. Currently, liver transplantation remains the only curative treatment for end-stage liver diseases; however, its application is severely limited by the global shortage of donor organs. To address this critical challenge, we aim to develop a highly efficient and safe cell-based therapy capable of treating liver diseases and related metabolic disorders in clinical settings.
Our cell therapy platform is based on chemically induced pluripotent stem cells (CiPSCs)—a next-generation, versatile technology for generating functional human cells and tissues (Science, 2013; Cell, 2025; Nature, 2022). The chemical reprogramming technology, developed by Hongkui Deng’s team at Changping Laboratory, provides an unprecedented opportunity to control somatic cell fate and generate desired cell types including pluripotent stem cells for applications in biomedicine in a highly flexible, efficient, and controllable manner. This reprogramming strategy mimics natural regeneration observed in lower organisms, sequentially reversing the developmental differentiation trajectory. Such a mechanism is fundamentally distinct from somatic cell nuclear transfer and transcription factor-mediated reprogramming. Unlike embryonic stem cells or transgene-induced pluripotent stem cells, CiPSCs eliminate ethical concerns, avoid transgene-associated safety risks, and offer a highly efficient, simple, and precisely controlled approach to reprogramming somatic cells. Remarkably, CiPSCs can be generated from just a single drop of fingerstick blood within 10 days (Nature Chemical Biology, 2025; Cell Stem Cell, 2025), making the technology both accessible and scalable.
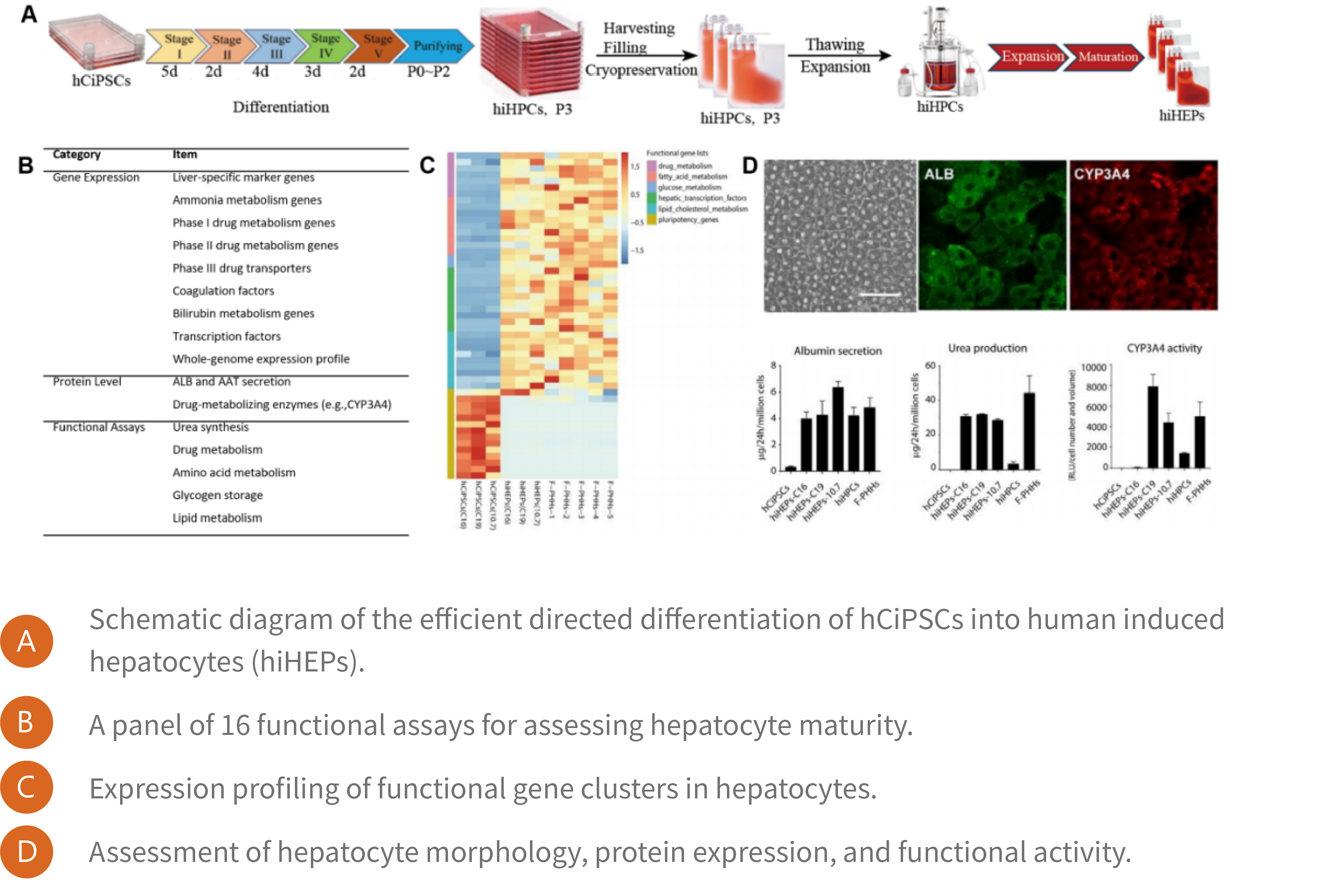
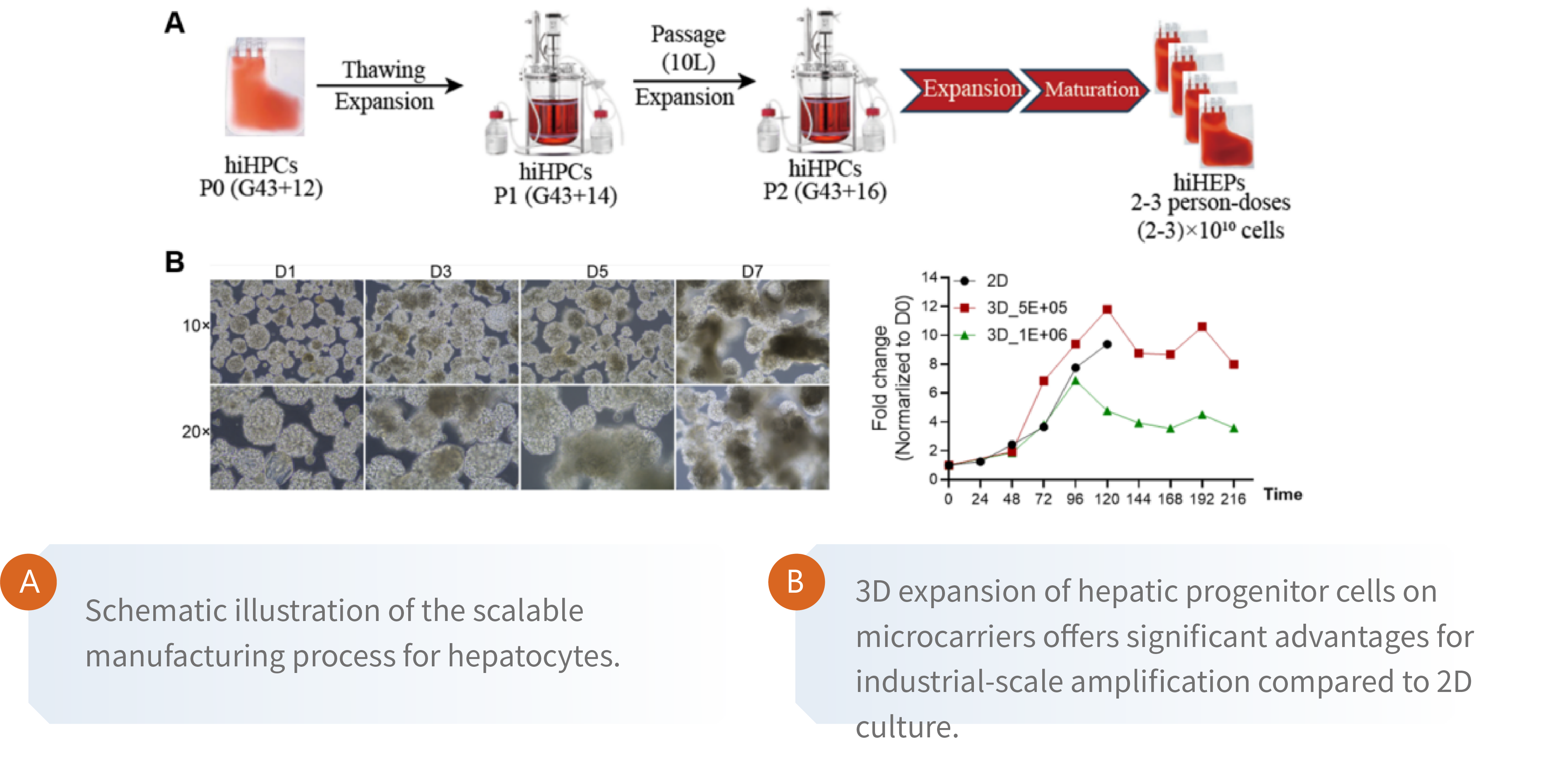
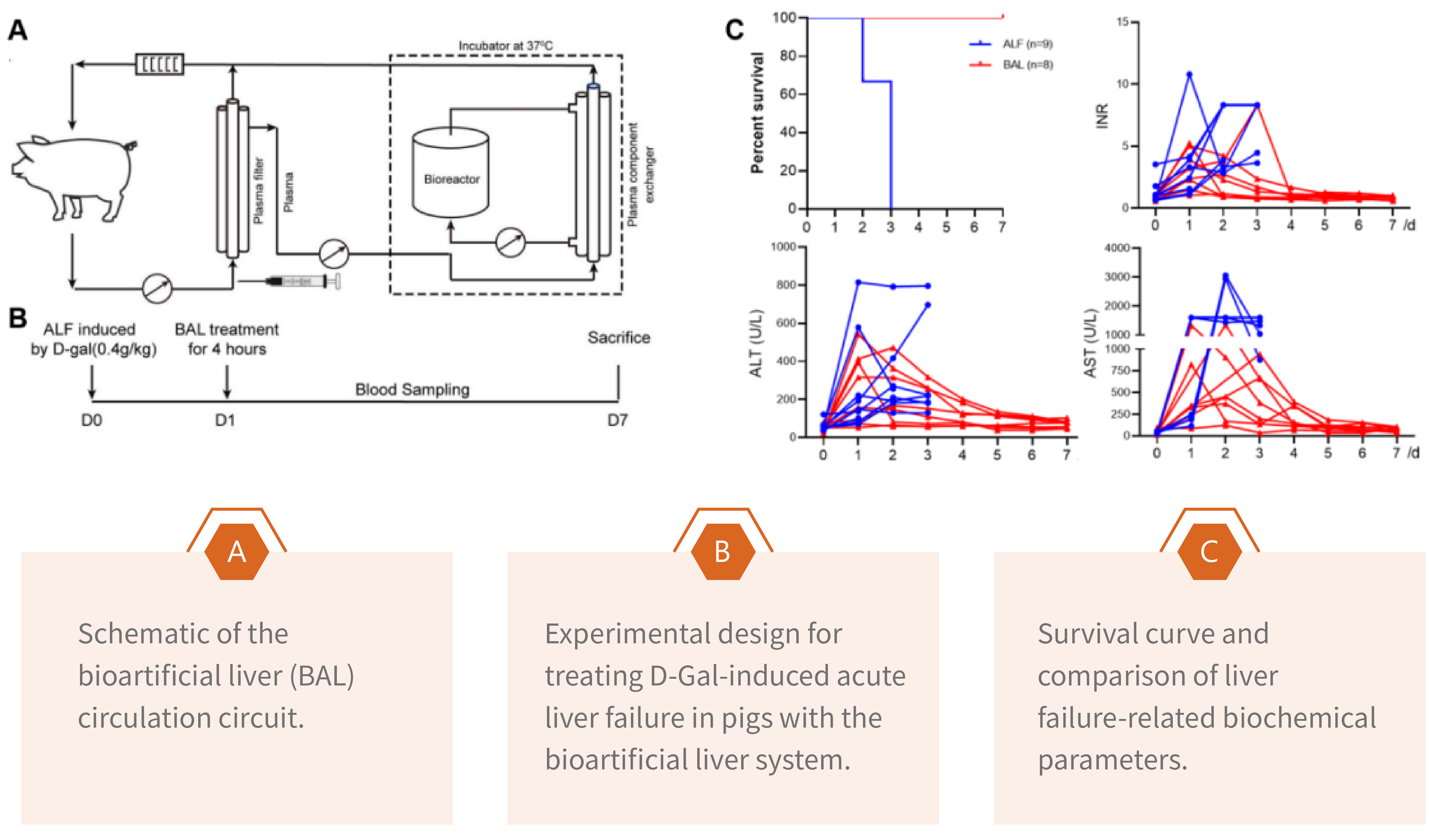
Using human CiPSCs, we have established a cost-effective 3D expansion system to produce large quantities of functionally mature hepatocytes (Hepatology, 2007; Cell Stem Cell, 2014; Science,2019; Cell research, 2020; Cell Proliferation,2023; Gut, 2025). These GMP-compliant, clinical-grade hepatocytes are utilized to construct an extracorporeal bioartificial liver (BAL) device that temporarily supports patients with acute liver failure, providing a bridge to either native liver recovery or liver transplantation. Remarkably, this bioartificial liver system has demonstrated a 100% success rate in reversing acute liver failure in pig models. To advance this groundbreaking technology toward clinical application, we are currently recruiting patients for an investigator-initiated trial (IIT) in hospitals.
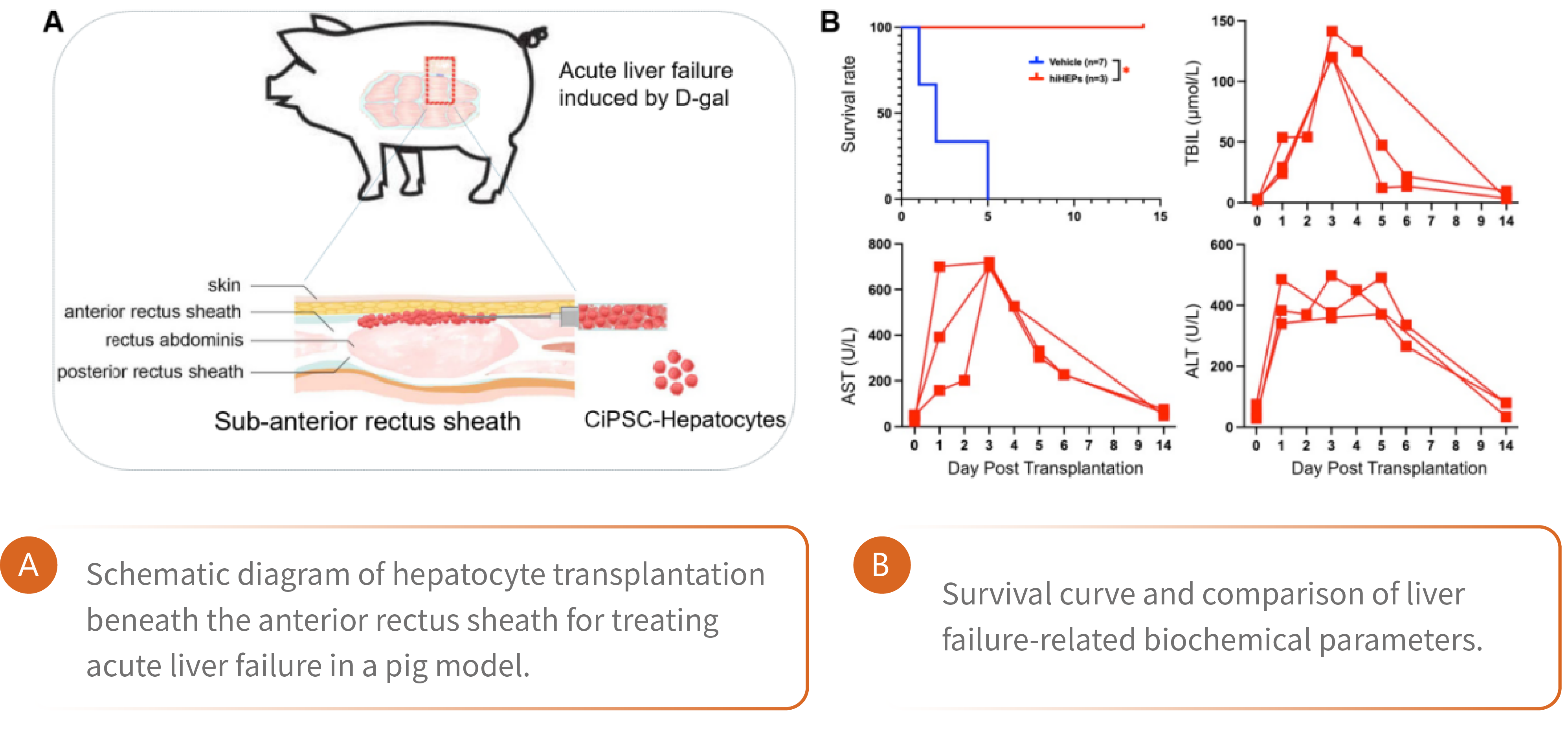
Beyond temporary liver support, we are also pioneering the transplantation of CiPSC-derived functional hepatocytes as a potential cure for chronic and end-stage liver diseases. This approach employs a novel transplantation strategy in which the cell grafts are implanted beneath the anterior rectus sheath of the abdomen. This site provides a favorable microenvironment that promotes the survival, maturation, and long-term maintenance of CiPSC-derived functional cells—a concept already validated in our clinical trials using CiPSC-derived islets to treat type 1 diabetes (Nature medicine, 2022; Cell, 2024; Cell stem Cell, 2025). In the near future, this strategy holds great promise to become a versatile and effective alternative to conventional liver transplantation, transforming the therapeutic landscape for liver disease management.
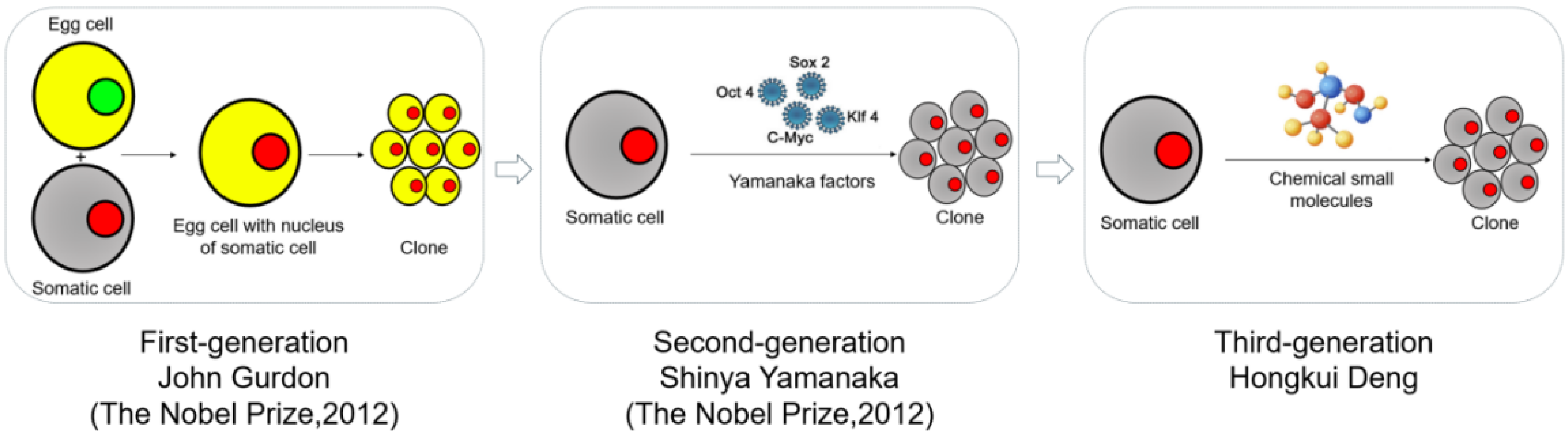
Chemical reprogramming represents the third-generation reprogramming technology, succeeding somatic-cell nuclear transfer and transgene-mediated reprogramming
Through 16 functional assays, CiPSCs-derived hepatocytes have been demonstrated to be comparable to adult primary hepatocytes
Scalable GMP-grade CiPSCs-derived hepatocytes enable clinical-grade bioartificial liver for liver failure
CiPSCs-derived bioartificial liver reverses liver failure in large animals, advancing to human trials
Innovative CiPSCs-derived hepatocyte transplantation: A potential alternative to liver transplants